History of batteries
From ancient relics to the contemporary power sources powering electric cars and international power systems, the history of batteries is a lengthy and intriguing one.
The most important turning points are shown in the following timeline:
Forerunners and Initial Findings
- Baghdad Battery (c. 3rd century BC): Archaeologists found clay jars filled with iron rods and copper cylinders, which some believe may have served as early batteries for electroplating.
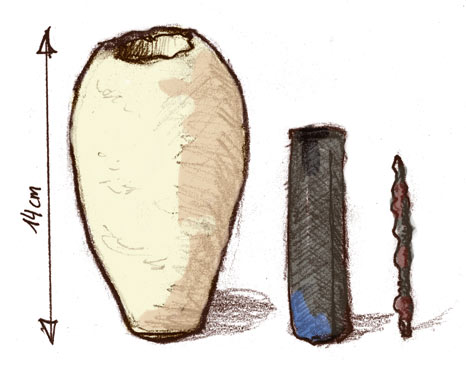
Figure 1, view larger image
- 1748: Inspired by the military phrase for a unit of artillery, Benjamin Franklin coined the term "battery" to refer to a collection of charged glass plates (Leyden jars) joined together.
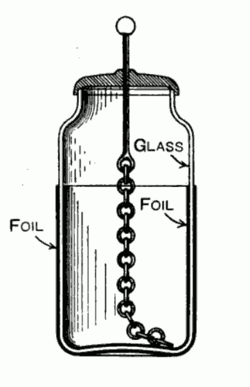
Figure 2, view larger image
- 1780: Animal Electricity: The foundation for comprehending the connection between chemical processes and electricity was laid by Italian biologist Luigi Galvani, who noticed that a dead frog's leg moved when touched by two different metals.
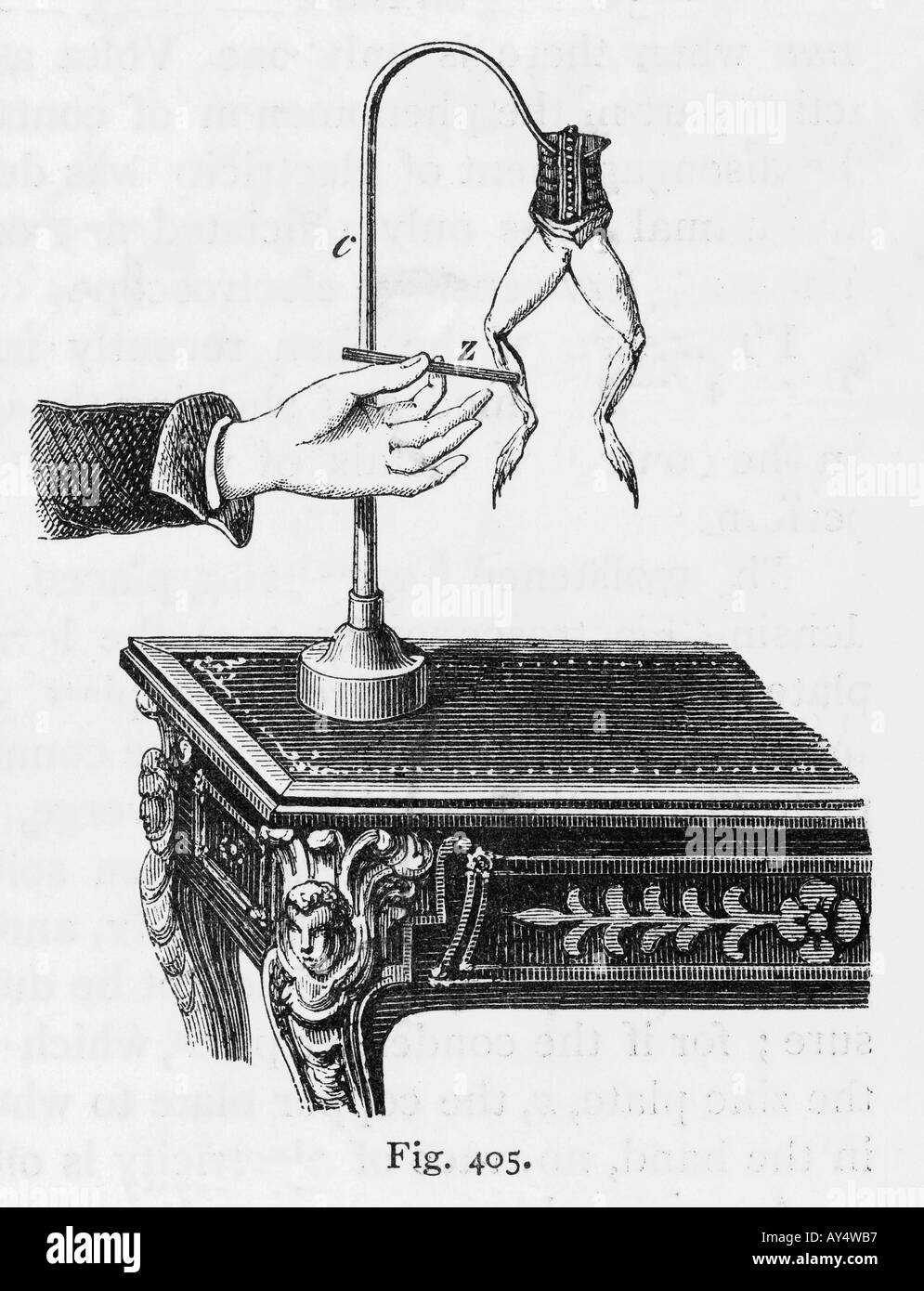
Figure 3, view larger image
The Original Real Batteries
- 1800: Alessandro Volta, an Italian physicist, created the Voltaic Pile, the first real battery. It was made up of alternating copper and zinc discs separated by brine-soaked paper or linen. Through a chemical reaction, this produced the first dependable, continuous electrical current source.

Figure 4, view larger image
- 1836 saw the invention of the Daniell cell by John Frederic Daniell, which solved the Voltaic Pile's rapid voltage drop (polarization) issue. The Daniell cell produced a more consistent, long-lasting current (about 1.1V) by utilizing two distinct electrolytes divided by a barrier. Telephones and telegraphs were frequently powered by it.

Figure 5, view larger image
The Age of Rechargeable Batteries
Figure 6, view larger image
Consumer Power and Dry Cells
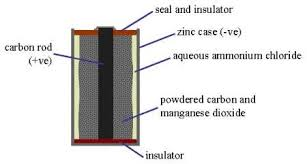
Figure 7, view larger image
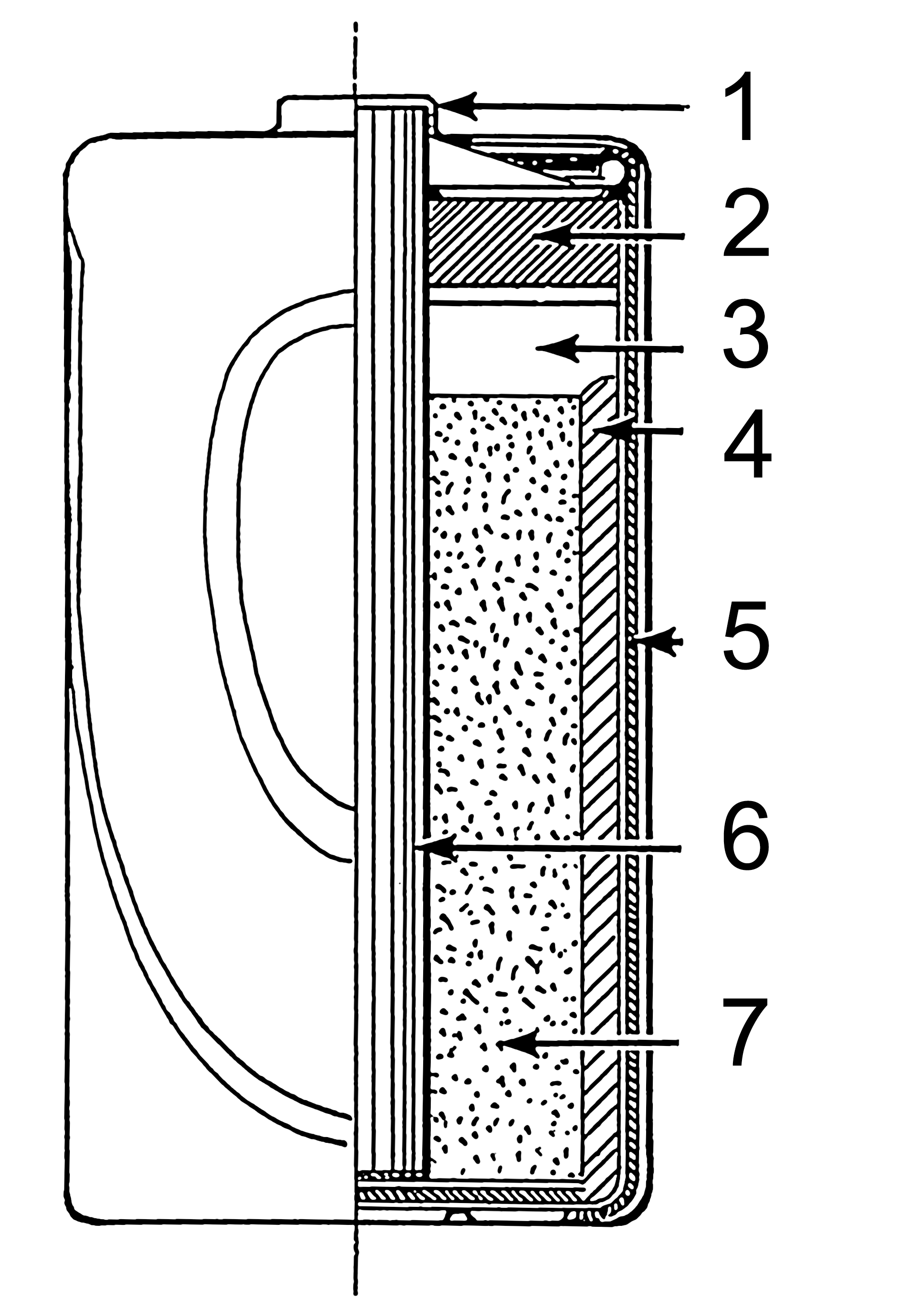
Figure 8, view larger image
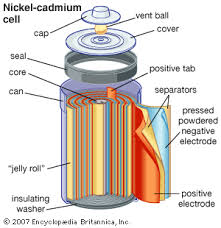
Figure 9, view larger image
Contemporary Batteries
- 1901: Nickel-Iron (Edison Cell): For use in electric cars, Thomas Edison created an alkaline storage battery.

Figure 10, view larger image
- 1955: Alkaline Battery: Lewis Urry, an Eveready employee, created the contemporary alkaline battery, which employed potassium hydroxide as the electrolyte. Compared to the earlier zinc-carbon cells, this chemistry allowed for a far longer life and a better energy density, swiftly establishing it as the industry standard for consumer electronics.
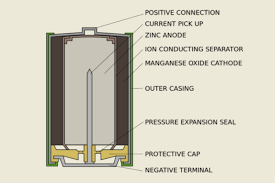
Figure 11, view larger image
- 1991: Lithium-ion (Li-ion): Sony introduced the first commercial Li-ion battery, building on decades of research. Li-ion technology was lighter, had a far higher energy density, and was not affected by the "memory effect" of NiCd batteries. The current revolutions in portable electronics and electric vehicles were sparked by this idea.
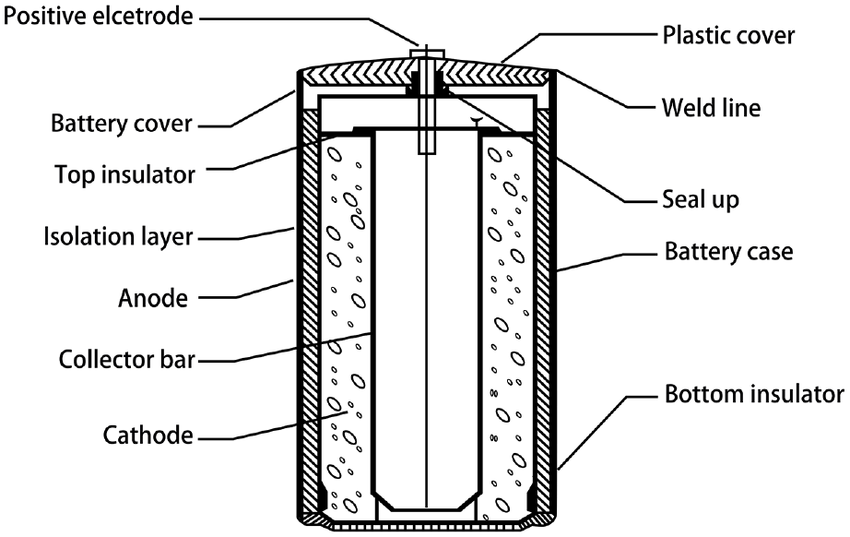
Figure 12, view larger image
thats all for this article
thank you for reading
AI has been used to rephrase Sentences

Figure 13, view larger image
Tech
89
4
Please sign in
Login and share
Sign in


























